Vascular access device (VAD) insertion is a common procedure in today's healthcare setting; yet, it remains one of the most invasive procedures undertaken, with 60% of patients in the UK and 80% in the US requiring some form of intravenous access (Lavery and Ingram, 2005; Hadaway, 2012; Wallis et al, 2014) (Table 1). Despite their common use, VADs continue to be associated with a high risk for infection and other complications. One complication that is emerging as a significant risk is medical adhesive-related skin injury (MARSI) (Farris et al, 2015). This type of injury is caused by a physical or mechanical reaction of the skin associated with the medical adhesive used in film dressings, tapes and adhesive fixation devices, particularly in the removal stage of their use. These injuries can be the catalyst for complications that can start at the catheter exit site under the dressing. A catheter exit site infection can remain localised to the soft tissue, or it can migrate into the bloodstream via the catheter (Inagaki and El Feghaly, 2019).
| Device | Indwell time | Indication | Vein access |
|---|---|---|---|
| Peripheral cannula | 4–10 days | IV therapy (non-vesicants) | Peripheral |
| Short midline (≤10 cm) | ≤29 days | IV therapy, blood sampling (non-vesicants) | Peripheral |
| Midline (≤25 cm) | 4–6 weeks | IV therapy (non-vesicants) | Peripheral |
| Acute non-tunnelled CVC | 10–14 days | All IV therapy, blood sampling | Central |
| Tunnelled cuffed CVC | ≥6 months | All IV therapy, blood sampling | Central |
| Peripherally inserted central catheter | ≥6 months | All IV therapy, blood sampling | Central |
| Implanted IV port | ≥6 months | All IV therapy, blood sampling | Central |
Source: Bodenham et al (2016); Royal College of Nursing (2016); Monreau and Alexandrou (2019). CVC=central venous catheter; IV=intravenous
Catheter-related bloodstream infections account for 10%–20% of hospital-acquired infections in the UK and are associated with both increased critical care unit admissions and mortality (Gahlot et al, 2014). Understanding the differences between intraluminal and extraluminal infection can inform the healthcare provider about which measures to take to avoid them occurring in the first instance (Kallen et al, 2010). In particular, the exit site carries a significant risk of bloodstream infections, as microorganisms find their way into the bloodstream through this site by means of biofilms that can grow on the outside surface of the indwelling catheter (Taylor and Palagiri, 2007; Han et al, 2010). Biofilms are produced by microorganisms on the catheter surface, protecting and nourishing the microorganism and making it difficult for antimicrobials to access them.
VAD-associated infections account for up to 20% of hospital-acquired infections in the UK, and are associated with increased length of hospital stay, mortality and treatment costs (Health Protection Agency, 2012). The National Institute for Health and Care Excellence (NICE) estimated the cost of treating a catheter-related bloodstream infection at approximately £9900 per case (Thokala et al, 2016). Plachouras et al (2018) reported that the European Centre for Disease Control (ECDC) estimated the cost of catheter-related bloodstream infections to European healthcare organisations to exceed €7 billion a year, with a single episode of catheter-related bloodstream infections costing from €4200 to €13 000. Although these data indicate the financial repercussions of catheter-associated bloodstream infections, they do not consider the long-term emotional and physical cost to the patient or the reputational damage to the healthcare organisation.
Bloodstream infections associated with VADs can be caused by numerous microorganisms, although there are some common culprits. Staphylococcus epidermidis, which can be found in the upper layer of the epidermis or mucosa, is considered normal flora on healthy skin (Zhou et al, 2020), but, if this microorganism finds its way into the body, especially the bloodstream, it can cause serious damage. S. epidermidis and the more serious Staphylococcus aureus are well established as major nosocomial pathogens (Meadows et al, 2009) associated with infections of indwelling medical devices, which spread via the formation of biofilms (Doulgeraki et al, 2017). Haymond (2019) classified bacterial biofilms as consisting of a slimy matrix on catheter surfaces, and the major virulence factor of these organisms is their ability to adhere to devices and form biofilms (Cerca et al, 2005). In particular, skin injured by a medical adhesive is vulnerable to infection via the catheter exit site.
When a VAD is coated with an S. aureus biofilm, the biofilm reaches the bloodstream, at which point a part of the biofilm can break off and travel to another part of the body, resulting in life-threatening infections, such as endocarditis; this is known as haematogenous spread (Grønnemose et al, 2017) (Figure 1).
IV catheter exit site infections are easy to recognise and treat if noticed in the initial stages. The site will show signs of phlebitis; redness of the skin is usually the first sign, with pain and swelling localised to the tissue surrounding the catheter exit site (Brooks, 2016). Intraluminal catheter infection is a more significant complication, and studies such as the one by Curran (2016) have shown that pathogens can find a way into the internal lumen of the IV catheter and then directly into the bloodstream, either because of colonisation of the needle-free connector (NFC) or an intraluminal biofilm. In both cases, poor practice during the manipulation of the NFC and injection of IV therapy may be the cause. Tissue adhesive injury can increase the risk of both types of infection; ultimately, a breakdown of good care and maintenance of the IV device is the main cause and is avoidable.
The introduction of care bundles is one strategy that has been shown to reduce the risk of infection in those needing VADs (Choi et al, 2013). A robust care bundle aims to standardise and validate vascular access practice across an entire medical organisation. Care bundles can prompt a number of specific episodes of care throughout a 24-hour period, which if undertaken, have been proven to reduce the risk of complications associated with VADs (Sichieri et al, 2018).
Care bundles are an established means of detecting signs of phlebitis but, unfortunately, using care bundles alone may not achieve a reduction in catheter-related bloodstream infections (Harnage, 2012), as each element of care requires the healthcare professional to know what action to take at the time of noticing a complication. The care bundle also relies on the healthcare professional undertaking the assessment in the first place, and non-compliance is a well-documented issue in the literature (Chopra and Shojania, 2013; McGuire, 2015; Simon et al, 2016). This should be taken into account when promoting care bundle use. Sadly, noncompliance with care and maintenance can often be the catalyst for more serious complications associated with vascular access, such as catheter-related bloodstream infections. This includes the removal of medical adhesive products (Table 2) from the catheter insertion site, which should always be a consideration in the care maintenance and maintenance of all VADs (Broadhurst et al, 2017).
| Type of device | Examples |
|---|---|
| Fixation/diagnostics |
|
| Stoma products |
|
| Adhesive wound dressings |
|
| Tapes |
|
| Wound tapes |
|
| Urethral catheter fixation and urinary sheath adhesive | |
Source: Fumarola et al, 2020
Evidence from a study by Frasca et al (2010) indicated that the most effective way to reduce the risk of infection is to ensure regular decontamination of the skin surrounding the exiting catheter and covering the device with a semipermeable, moisture-control dressing, which ensures that the decontaminated area of skin is larger than the film dressing being used to cover it. Once in place, the dressing should be left undisturbed for 7 days, unless debris is seen under the dressing or the dressing starts to come away from the skin (Royal College of Nursing, 2016). The use of medical adhesive removal products should be considered a priority for the removal of film dressings and other adhesive fixation devices, as these are the most common cause of skin injury, particularly for peripherally inserted central catheters (PICCs) (Broadhurst et al, 2017).
When adhesive tapes or dressings are removed from unprotected skin, varying amounts of the outermost layer of the skin, consisting of keratinised cells, are removed. The repeated application and removal of adhesives from the same skin site can cause exogenous damage (skin stripping) or MARSI (Table 3). Repeated injuries of this type may then result in inflammatory skin reactions, local oedema and soreness, all of which can have a further adverse effect on the skin's barrier function and lead to local or systemic infection (Dykes et al, 2001; Cutting, 2008; McNichol and Bianchi, 2016).
| Type | Cause | Result | Example |
|---|---|---|---|
| Mechanical | Skin stripping | Removal of adhesive tape or dressing | Removal of one or more layers of the epidermis (top layer of skin). Damage is often shallow and irregular in shape. The skin may appear shiny. Open sores may be accompanied by red skin and blister formation |
| Tension injury or blister |
|
Injury caused by shear force (separation of the epidermis from the dermis, which is the second layer of the skin) | |
| Skin tear |
|
Skin is pulled away and the layers of the skin separate. This can either cause a partial-thickness wound (a wound that extends into the epidermis and dermis), or a full-thickness wound (which extends into fat and muscle layers) | |
| Dermatitis | Irritant contact dermatitis | Response to contact with a chemical irritant in the adhesive | Skin is inflamed (red), and can become blistered, dry, thickened and cracked |
| Other | Maceration | Skin damage resulting from prolonged accumulation of moisture under an adhesive tape or dressing | Skin appears wrinkled and white/grey. Softening of skin increases permeability, and susceptibility to infection |
| Folliculitis | Prolonged accumulation of moisture and heat under an adhesive can attract bacteria, which might proliferate in this enclosed environment, resulting in an inflammatory response in the hair follicle | Appears as small inflamed elevations of skin around the hair follicle. These can present as papules (skin that has changed colour or texture) or pustules |
Source: Fumarola et al, 2020
Collier (2004), Dryden et al (2015) and Reevell et al (2016) indicated that any compromise in skin integrity caused by medical adhesive injury can lead to clinical complications. The evidence base points to the use of sterile skin protectants or sterile adhesive removers in order to reduce the risk of these complications.
MARSI can be common when associated with the care and maintenance of VADs, especially due to the long-term nature of some of the catheters that are used. The repeated trauma to the skin due to the adhesive can have a huge impact on patient experience and make weekly care and maintenance of the catheter an ordeal for the patient (Hitchcock and Savine, 2017). Some patients are at higher risk than others. Older people are at particular risk of injury because their skin is fragile. Collier (2019) outlined the changes to older people's skin, which include: a reduction in dermal thickness and subcutaneous fat, delayed healing, damage due to ultraviolet radiation from the sun over time, and reduced blood supply. Other conditions such as diabetes and those causing peripheral oedema and dermatitis are also risk factors.
A comprehensive and evidence-based consensus document on the assessment and prevention of MARSI (Fumarola et al, 2020) covers all forms of medical adhesives in clinical practice and is highly relevant to vascular access and IV therapy practice. The guidance outlines the definition of MARSI and provides an overview, part of which states that a MARSI is a term used to define any skin damage related to the use of medical adhesive products or devices. The guidance states that this type of injury is largely avoidable. The overall consensus is that MARSI can occur when the attachment between the skin and an adhesive is stronger than that between individual cells, causing either the epidermal layers to separate or the epidermis to separate completely from the dermis (mechanical trauma) (Table 4).
| Intrinsic risk factors | Extrinsic risk factors |
|---|---|
|
|
|
Source: Fumarola et al, 2020
The application of medical adhesive removers in vascular access is becoming more common, with the advent of vessel health and preservation (Fiorini et al, 2019), and skin preservation is also becoming a priority. Choosing the best product to use to achieve safe and effective removal of medical adhesive is important, because not all products have the same properties.
There are many medical adhesive remover products available on the market. Most are focused on stoma care. Removal of stoma adhesives requires larger volumes of product to be repeatedly applied to the stoma site. These products are often supplied in a traditional aerosol spray can, which is not sterile. Products used on and around VADs should be sterile, as this will reduce the risk of infection. A leading brand of medical adhesive remover that is produced in a sterile form is Appeel Sterile manufactured by CliniMed Ltd, and the present article focuses on this product.
Appeel Sterile medical adhesive remover has been available for over 10 years, and its benefits have been previously documented (Stephen-Haynes, 2008; Cooper, 2010; Adderley, 2018; Evans, 2019). Appeel Sterile is a silicone-based, no-sting medical adhesive remover that removes medical adhesive easily and painlessly. It is gentle enough to use on sensitive skin, for example, in children and older people with fragile skin. Appeel Sterile dries in seconds and will not affect the re-adhesion of a replacement film dressing and adhesive fixation device.
Appeel Sterile is available in a spray can, foam applicator, wipe or as a liquid sachet. This range of product choice gives health professionals options for using the product depending on the size and location of the adhesive product requiring removal. The following case studies show the many different applications for the use of Appeel Sterile in vascular access practice.
Case study 1
An 80-year-old woman had septic arthritis (Figure 2). She had a history of chronic obstructive pulmonary disorder and type II diabetes. She was an inpatient receiving intravenous antibiotics and corticosteroids, and had a 24-gauge peripheral cannula on the left dorsum of the hand that had been in situ for 96 hours.
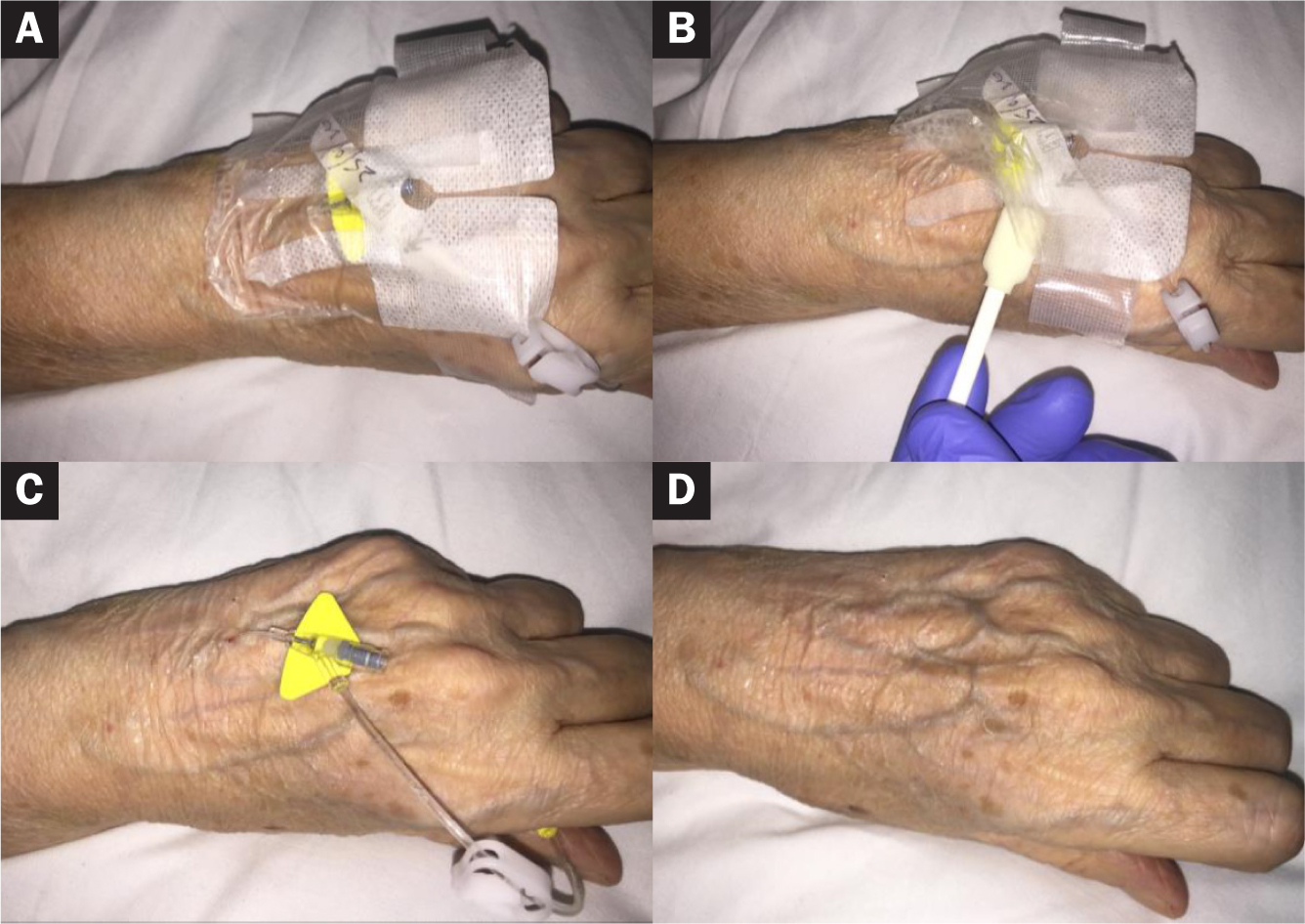
The peripheral cannula was inserted and secured with an appropriate semipermeable IV dressing and adhesive securement strips in line with organisational guidelines.
The patient's skin was fragile due to her age and condition, and this was exacerbated by the use of steroids. The patient was anxious about having the cannula taken out because she felt her skin was very sensitive to the film dressing being removed. She reported experiencing pain and discomfort when the last film dressing was removed from a previous peripheral cannula.
The Appeel Sterile medical adhesive remover foam applicator was used to remove the IV dressing and securement strips without any complication or noticeable trauma to the skin. More importantly, the patient did not experience any pain or discomfort. After removal of the film dressing and peripheral cannula, there was no noticeable redness or irritation to the skin.
The foam applicator was easy to use to apply Appeel Sterile under the corner of the dressing first, and it was then worked under the entire film dressing.
Case study 2
A 41-year-old man had lower limb cellulitis (Figure 3). He was being treated in ambulatory care with intravenous antibiotics administered at home via a 3Fr midline catheter, which was due to be removed after 2 weeks of therapy. The patient experienced pain and sensitivity during dressing and fixation device removal, because the area being treated was above the antecubital fossa where there was hair growth.
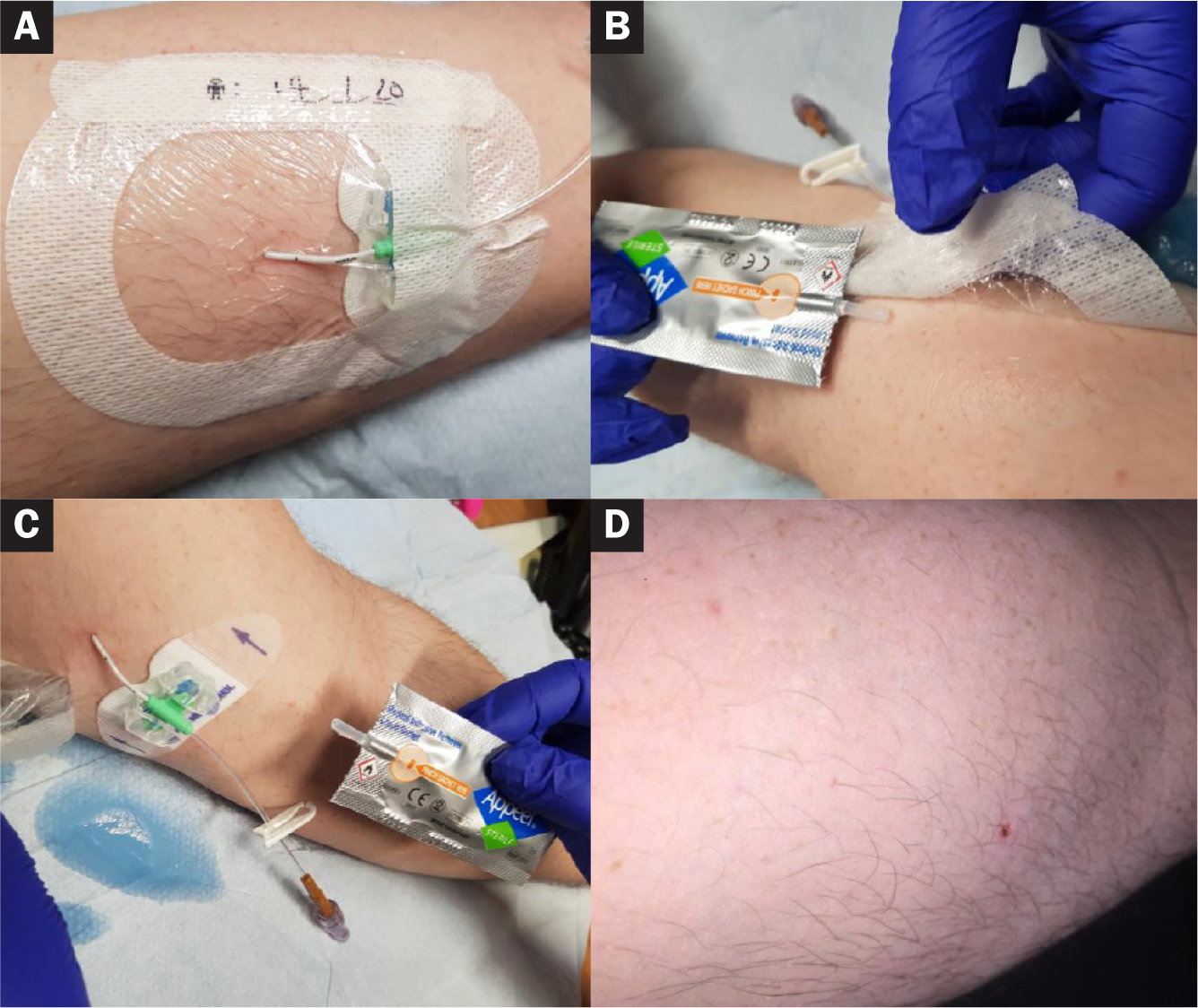
The midline catheter was dressed with an intravenous, semipermeable film dressing and held in place with an adhesive foam locking device. Both these were changed after 7 days, in line with local guidelines. The midline catheter was removed 7 days later, along with the film dressing and fixation device, using the Appeel Sterile liquid sachet.
The patient reported no pain or discomfort during the removal of the medical adhesive products, and the skin and hair under the film dressing remained intact without any injury.
The Appeel Sterile liquid sachet includes a fixed application straw inside the packet which makes it very easy to apply the liquid in a controlled manner on and under the medical adhesive products. The liquid completely removes all medical adhesive and leaves no residue after use.
Case study 3
A 9-year-old boy experienced acute exacerbation of asthma (Figure 4). He was admitted for intravenous corticosteroids, antibiotics and fluids. The cannula was in situ for 4 days and was removed in order to discharge the child home.

The patient was previously admitted for treatment for the same condition and was well-known to the IV team. He had a fear of needles and the associated removal of the peripheral cannula, partly due to the discomfort experienced during film dressing removal. This was the first time a medical adhesive remover was used on the patient to remove the film cannula dressing, and it made such a difference to him that his parents were given an Appeel Sterile Foam Applicator to take home and bring with them the next time their child needed to be admitted and a cannula needed removal. The Appeel Sterile Foam Applicator easily removed the film dressing, and the process was painless. There was no adhesive residue left after removal.
Case study 4
An 84-year-old woman attended the IV day unit for a blood transfusion via a peripheral cannula (Figure 5). The patient was being treated for aplastic anaemia and was receiving regular blood transfusions as a day patient.
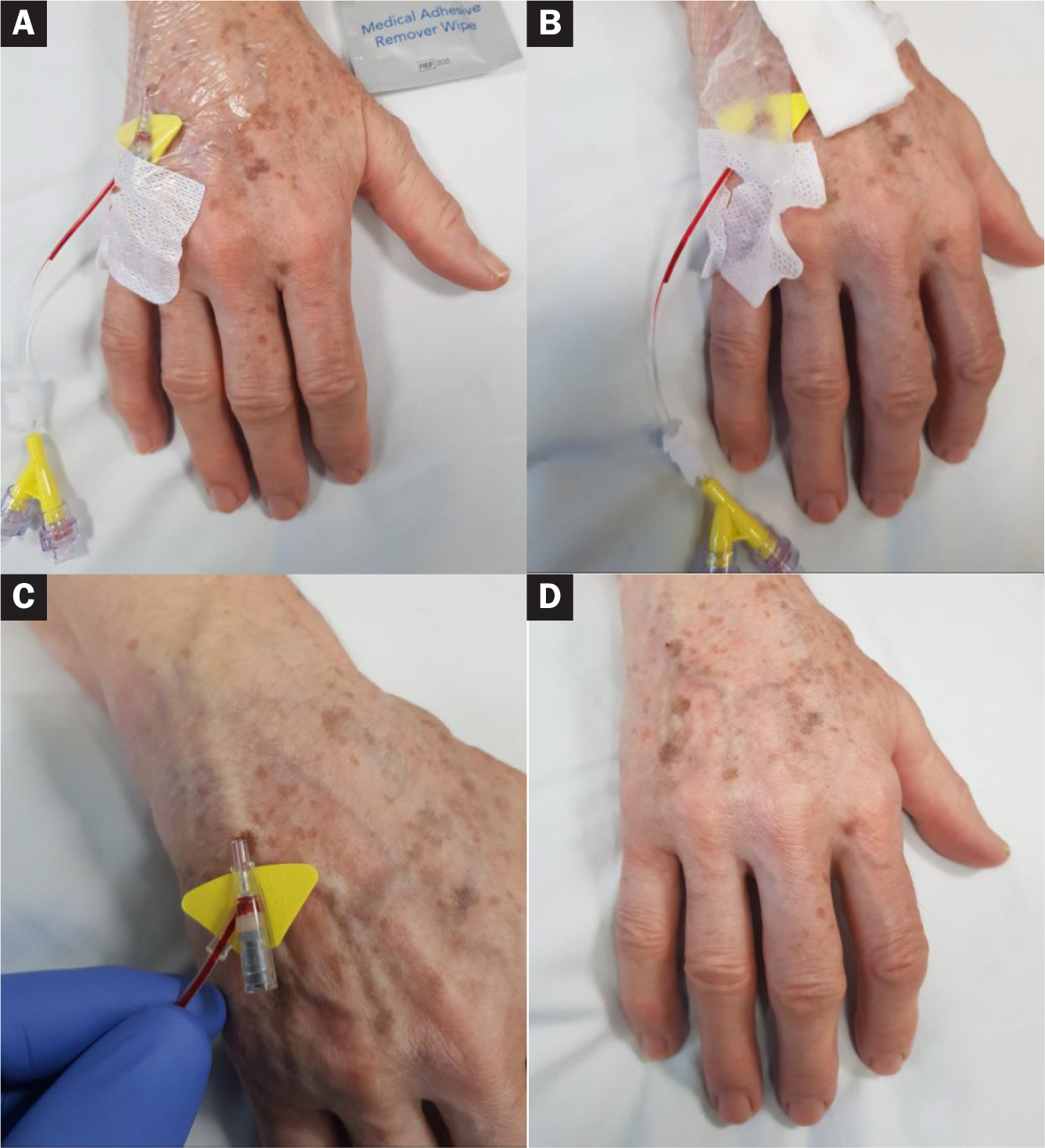
The patient had fragile skin, which was prone to injury from the medical adhesive during cannula removal. The patient had undergone numerous peripheral cannulations previously and was used to experiencing pain and discomfort during cannula removal. The use of Appeel Sterile medical adhesive remover in the form of an impregnated wipe enabled the cannula film dressing to be removed easily with no pain or damage to the surrounding fragile skin.
This was the first time the patient has experienced the use of a medical adhesive remover, and she was extremely happy with the outcome. She has requested that Appeel Sterile is used the next time a cannula needs to be removed.
Conclusion
There are many complications associated with VADs that can be significantly reduced if evidence-based care and maintenance practice are followed. It is clear from the evidence that the risk of these complications can be reduced through the use of care bundles. The removal of VADs needs to become part of the care bundle to ensure that MARSI is an aspect that is regularly risk assessed for; this should initiate a standardised approach to prevention and treatment if an injury should occur. Avoidance is better than treatment after the occurrence. The risk of every patient experiencing a MARSI should be considered, and a medical adhesive remover should be used.
Appeel Sterile is an effective product that is sterile and, as shown by the presented case studies, can perform exceptionally well in removing adhesive film dressings and fixation devices with no pain, discomfort or skin damage.
The routine use of Appeel Sterile should, therefore, be considered when removing medical adhesive products in vascular access to assist in maintaining vessel health and preservation.

