The Gram-negative bacterium Acinetobacter baumannii can survive on solid and dry surfaces for up to 5 months and shows a high level of innate and acquired antimicrobial resistance (Nowak and Paluchowska, 2016). Multidrug-resistant Acinetobacter baumannii (MDR-Ab) is an important cause of healthcare infections (Doi et al, 2015; Singh et al, 2015; Nowak and Paluchowska, 2016). It is capable of causing both community- and hospital-acquired infections (HAIs), although HAIs are the most common form (Fournier et al, 2006) and few strains have been recovered from community sources (Antunes et al, 2014).
Recognised as one of the six most important multidrug-resistant microorganisms (MDROs) in hospitals worldwide (Antunes et al, 2014), this pathogen is responsible for a vast array of infections, of which ventilator-associated pneumonia and bloodstream infections are the most common. Mortality rates can reach 35% (Antunes et al, 2014).
It can enter the body readily through open wounds, intravascular catheters and mechanical ventilators (Antunes et al, 2014). Patients who are immunocompromised, such as those being nursed in intensive care, neonatal intensive care and burns units, are particularly vulnerable to Acinetobacter baumannii infections, because of their serious underlying conditions that may require the increased use of highly selective antibiotics, indwelling lines and other invasive devices (Antunes et al, 2014).
Antimicrobial resistance has progressively increased since the 1970s, at which time the majority of strains were sensitive to commonly used antibiotics (Antunes et al, 2014). In recent decades Acinetobacter baumannii has acquired resistance to many commonly used antibiotics (Antunes et al, 2014).
Environmental contamination of hospital items with Acinetobacter baumannii has been identified as an important and persistent cause of cross-transmission, with contaminated items including suction equipment, pillows, mattresses, sinks and wash hand basins (Fournier, 2006). Investigators showed that Acinetobacter baumannii strains could be isolated from a hospital bed rail 9 days after the discharge of an infected patient from hospital, indicating the lengthy survival times of the organism (Fournier, 2006). In some conditions it was found that Acinetobacter baumannii organisms are able to survive for an average of 20 days (Fournier, 2006).
Studies have confirmed that the survival of Acinetobacter baumannii in the environment could contribute to the transmission of the organism during outbreaks (Fournier, 2006; Espinal et al, 2012). At the time of an outbreak in two wards in one NHS foundation trust in 2015, the authors found no data more current than that provided by Fournier (2006), indicating a clinical knowledge gap about the impact of Acinetobacter baumannii.
This article examines the management of an outbreak of MDR-Ab, which affected two adjoining respiratory medicine wards; one with 30 beds, the other with 28. The outbreak was continuous from April 2015 to January 2016, followed by two further cases in July and August 2016. There were six male and six female patients affected whose ages ranged from 37 to 83 years, with a median age of 72. The first patient identified, whose symptoms indicated the existence of the outbreak, is referred to throughout this paper as the ‘index case’ in line with infectious diseases terminology.
Having been previously cared for in a healthcare establishment in a developing country, following a stroke, the index case was repatriated to an acute NHS trust in the north east of England. Four weeks after admission the patient was found to be colonised with MDR-Ab, resistant to meropenem and sensitive only to colistin (an antibiotic of last resort). Isolation is recommended for the control of an MDR-Ab outbreak (Cookson et al, 2006), however, the patient had complex needs which included neurological and mental disability, meaning that constant observation was required.
The outbreak was provoked by failure to isolate the index case both before and after the first identification of MDR-Ab in urine and sputum specimens. Principal outbreak investigation findings/lessons learned indicated that the decision not to isolate on first notification of the positive result was based on the patient's dependency level and the need to be constantly observed in the bay. There was discussion with the consultant physician around the risk to the patient's airway if not under constant visual and auditory supervision, the need for 24-hour registered nurse care and difficulty with manual handling, which required six staff (due to the presence of several invasive devices). Staff may not have recognised the gravity of the possible transmission of MDR-Ab on the unit as it was not a common pathogen. This lack of awareness adversely affected the implementation of infection prevention and control (IPC) precautions.
The patient was accommodated in a bay where he was visible at all times and where nursing duties could be undertaken by more than one nurse. Control measures to mitigate the risk to other patients included the use of standard IPC precautions, barrier nursing within the bay area and enhanced levels of cleaning of equipment and the environment.
The index case remained in an open bay, alongside other patients, for 6 weeks before being placed in isolation. During this time the patient was inhaling his own secretions repeatedly, which caused vigorous coughing episodes, meaning that the colonised respiratory secretions were a potential source of transmission both directly through colonised droplets and also via airborne dissemination (Wainwright et al, 2009). Efforts were made to manage this risk through staff use of filtering facepiece respirators (FFP3 masks) and visors.
Stringent efforts, such as environmental cleaning with a chlorine-releasing agent at increased frequency and the use of enhanced personal protective equipment (PPE), were made to avoid cross-transmission, although some contamination of the environment was inevitable. Despite these measures, other patients on the adjoining respiratory ward were subsequently found to be positive for the same organism. In total, by the end of August 2016, four patients had shown signs of clinical infection and a further eight had been revealed to be colonised with the bacterium. The initial patients were identified from clinical specimens, but once a programme of patient contact screening was implemented in April 2015, identification was also provided from screening.
At the time of the outbreak, a literature review revealed no guidelines for the decolonisation of MDR-Ab cases specifically and identified a single study that assessed the effectiveness of decolonisation regimens. The study, which was performed between 2002 and 2003, suggested that whole-body disinfection with 4% chlorhexidine gluconate daily until discharge significantly reduced Acinetobacter baumannii skin colonisation (Borer et al, 2007).
The trust's MDR-Ab microbiological testing did not show antibacterial action with chlorhexidine but octenidine did have an antibacterial effect. The trust was already using octenidine to decolonise methicillin-resistant staphylococcus (MRSA)-positive patients, hence the decision to decolonise with octenidine rather than chlorhexidine.
Outbreak measures utilised
Investigations
Environmental screening was not initiated until there was evidence of spread to another patient. This was because some contamination of the immediate environment was considered inevitable and measures had been put in place to mitigate this risk. In July 2015 environmental screening commenced. Advice was sought from expert microbiologists on the optimum method to obtain samples, and on laboratory testing protocols.
Testing of the environment was performed using hygiene swab sampling kits (Enviroscreen sponges; Technical Service Consultants Ltd) in the ward managing the index case and the adjoining ward. Samples were taken from a number of sites, including patient equipment, furniture, wash hand basins and case note folders/trolleys.
The environmental sites were selected based on common sites identified in published accounts of other outbreaks (Thom et al, 2011). The sampled sites were not in close proximity to the index case, who was being nursed in isolation at the time of environmental swabbing. The aim of the swabbing was to determine whether there were any environmental reservoirs through which the organism could spread between patients.
In total, over 300 samples were obtained, eight of which were positive for MDR-Ab. It was isolated from a wash hand basin in the side room of a known affected patient. The other items of equipment from which MDR-Ab was isolated included a hoist, walking frames, notes trolleys, a blood pressure monitor and a Rota Stand (used to help transfer a patient from one position to another).
Although environmental screening was conducted on a further 14 occasions between August 2015 and September 2016, no other environmental samples proved positive.
Contact tracing (active case finding to increase the rate of identification) began at the end of April 2015 following discussions with the Public Health England (PHE) team. This was restricted to hospital contacts who remained as inpatients. At this point an additional patient to the index case was found to be positive for MDR-Ab.
Initially, patients who had been nursed within the same open area as the index case and who remained in hospital were screened. In May 2015, as a one-off exercise, screening was extended to all inpatients on the two adjoining wards. As new cases emerged, inpatients who were found to have had contact with a patient colonised or infected with MDR-Ab, were also screened.
Screening was then repeated weekly or until the patient was discharged or had three negative screens. Patients were not followed up in the community if discharged to their own home, unless they sought this from their GP. Patients discharged to nursing or residential care were followed up on a case-by-case basis with input from the IPC nurse for care homes and the IPC doctor. Patient screening specimens were nasal and groin swabs, swabs from any wounds or invasive device (e.g. percutaneous endoscopic gastrostomy (PEG) tubes, tracheostomy tubes, and IV lines), urine, a sputum sample if expectorating and either a stool sample or rectal swab.
All staff on both wards were screened including nurses, doctors, domestic staff and physiotherapists. For some staff the screening was repeated a second time. The screening was initiated in early May 2015 on the advice of the IPC doctor. The total number of staff screening samples processed was 256; all were negative. Staff screening consisted of nasal and skin lesion swabs. In addition, a staff hand plating exercise was undertaken to detect transient carriage but none was found; this involved staff pressing their hands onto agar plates, which were then cultured in the microbiology laboratory.
Control
Cleaning with chlorine was implemented when the index case was identified. This was initially restricted to the immediate environment around the patient but was then increased to the ward areas as further cases were identified. At the beginning of the outbreak, chlorine was used daily at a concentration of 10 000 parts per million (ppm) for 4 weeks. This was then reduced to 5000 ppm for cleaning and wash hand basin flushing in infected rooms. Wash hand basins were flushed daily with the chlorine concentrate, which remained in the trap of the drainage outlet until the basin was next used. The product manufacturers recommend 10 000 ppm for blood spillages and 1000 ppm for the general clinical environment. At the outset it was agreed to use the product at its maximum concentration but as the situation was brought under control it was felt that a strength of 5000 ppm would be sufficient to control transmission.
The trust had already agreed use of hydrogen peroxide vapour (HPV) ‘fogging’ as its gold-standard approach to room decontamination following environmental exposure to Clostridium difficile (Boyce, 2008). This decision, a multidisciplinary one, made several years prior to the outbreak, was agreed by nursing, IPC, microbiology and estates facilities departments as a result of having tested systems on the market for efficacy, safety and practicality. As a result the trust responded to the MDR-Ab outbreak with this gold-standard approach. The ward hygienist team was responsible for the deep cleaning of side rooms/bays and then carried out bio-decontamination with exposure to HPV fogging.
Environmental bio-decontamination with HPV fogging was ongoing across both wards throughout May and June 2015. As it was not possible to close an entire ward, decontamination of individual patient bays was achieved by closing one multi-occupancy bay and one single room per ward. Once decontaminated, these areas were used for rotation of the remaining patients until all patient rooms had been treated. Some areas, including utility rooms, offices and corridors, were not treated as they remained in continual use.
A 12% HPV was used for the environmental fogging and 35% for the equipment chamber. Ward areas were treated using mobile HPV machines and all patient equipment was processed through the trust's onsite hydrogen peroxide gas chamber facility operated by a dedicated team of hygienists.
Some of the procedures subsequently outlined in the guidelines (Wilson et al, 2016) were used, including the use of disposable PPE, hand hygiene, environmental screening, decontamination of respiratory equipment, terminal decontamination with hypochlorite, use of HPV and monitoring of staff hand hygiene. These guidelines were not published at the time of the documented outbreak.
Appropriate PPE was in use for nursing the index case from the outset. When the second case was identified on 29 April 2015, an outbreak was declared and enhanced PPE, including long-sleeved gowns, FFP3 masks and face visors were brought into use.
Over the period of the outbreak patients who tested positive for MDR-Ab were isolated immediately and staff undertook barrier precautions. Hand-hygiene practices were reinforced, including the use of wash hand basins for this purpose only. IPC practices on both affected wards were reviewed daily.
Decolonisation
Owing to a lack of published data at the time of the outbreak, decolonisation products containing octenidine (schülke UK Ltd) and chlorhexidine underwent microbiological testing in the hospital laboratory to assess efficacy before use. As octenidine, showed antibacterial action against MDR-Ab, individually tailored decolonisation regimens were introduced for colonised and infected patients. These included the administration of products containing octenidine for 7 days (following the manufacturer's instructions). The octenidine decolonisation regimen comprised:
Patients who showed clinical signs and symptoms of infection also received 7 days' treatment with the antibiotics teicoplanin, rifampicin and intravenous and/or nebulised colistin, according to trust protocol. Following decolonisation and any treatment, patients were left untreated for 2 days. Screening was then repeated.
Results of the measures taken
Key dates in the outbreak are shown in Figure 1. The outbreak lasted 9 months from April 2015 to January 2016 and was declared closed on 12 January 2016. However, a further two cases were identified in July and August 2016. The first of these later cases was thought to have acquired the organism during the initial outbreak. It was speculated that the second case of 2016 may have been caused by transient carriage on the hands of healthcare workers (although there was no direct evidence for this). Regular hand-hygiene audits were undertaken and scores for the two wards ranged between 73% and 100% throughout June and July 2016.
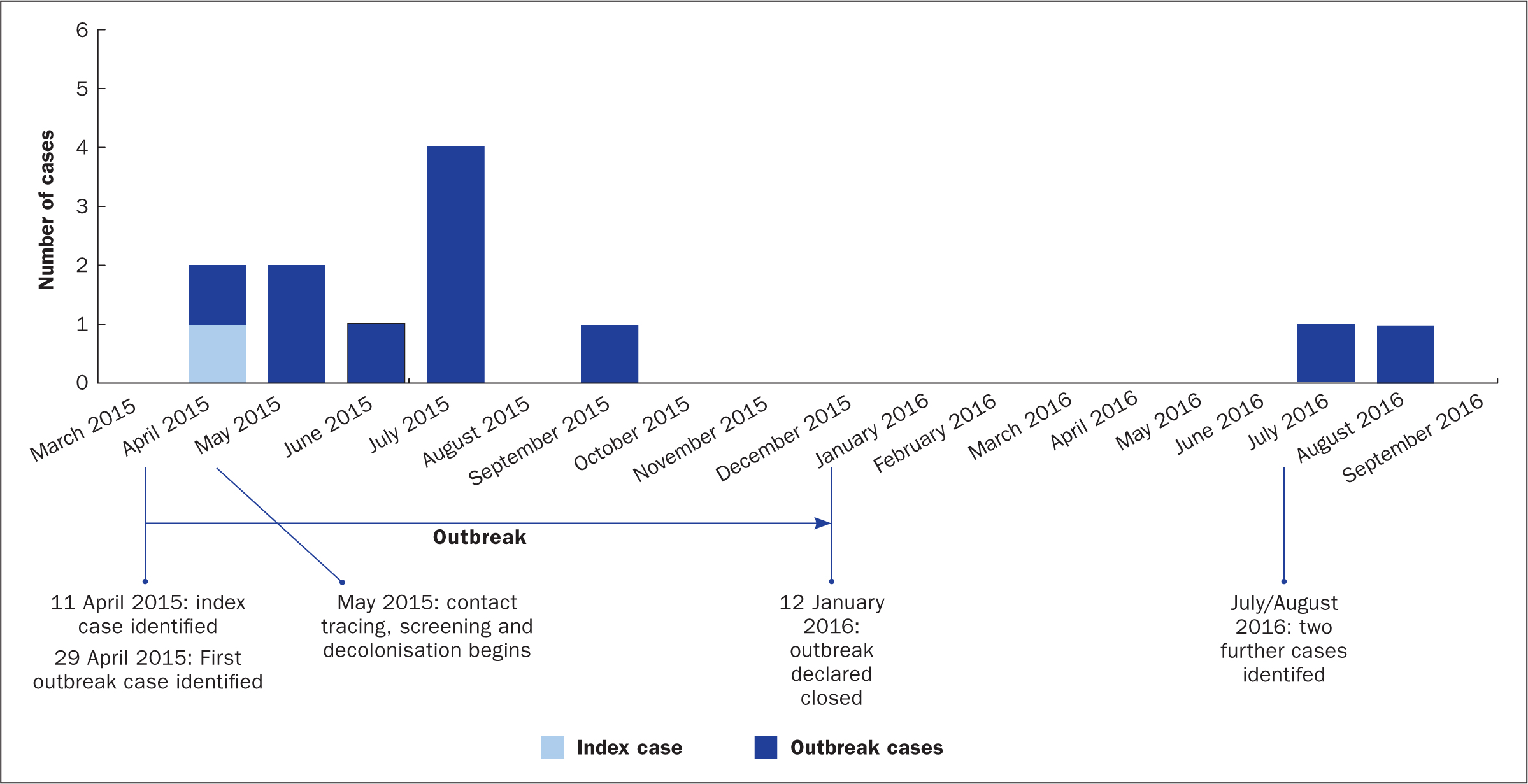
Transient carriage of organisms on the hands of healthcare workers is well documented and hand contamination despite wearing gloves has been reported in 4.5% of healthcare workers after caring for a patient with MDR-Ab (Morgan et al, 2010). Although it is not possible to be certain that this led to the second case in 2016, and environmental or equipment factors cannot be entirely ruled out, the authors believe that transmission by healthcare workers' hands was the most likely cause, particularly as by virtue of its transience MDR-Ab may not always be revealed by staff hand plating. There were no further cases and repeat environmental screening (93 samples) failed to produce any positive MDR-Ab results.
Overall, 12 patients across two adjoining wards (10 in 2015 and two in 2016) tested positive for MDR-Ab. Four of these showed signs and symptoms consistent with clinical respiratory infection, the remainder being colonised.
The index case was rescreened following initial decolonisation and treatment with nebulised and intravenous colistin. Although the patient did not have a completely negative screen, some sites were successfully decolonised. Screens were not always full/complete, i.e. did not include specimens from all required sites or were, on occasion, rejected for failing to meet laboratory criteria. Between May and September 2015 the following sites each returned some positive and some negative results: nose, groin, urine, catheter site, rectum, sputum, tracheostomy and PEG. All screening from 10 September onwards returned negative results including the final screening collected on 27 November 2015 which was negative in nose, groin, rectum and PEG. Re-colonisation of these sites with MDR-Ab occurred at different stages throughout his admission. Similarly, other colonised and/or infected patients were intermittently negative on screening after treatment cycles with nebulised, oral and intravenous colistin and/or the octenidine decolonisation regime.
Although MDR-Ab-positive patients were found to be intermittently positive, the use of octenidine may have reduced the transmission of MDR-Ab between patients and therefore reduced the number of positive cases. Further work is needed to investigate the role of octenidine in the management of an MDR-Ab outbreak.
Environmental screening was undertaken during the outbreak with over 300 samples being obtained. Eight samples were positive. MDR-Ab was isolated from a wash hand basin in the room of a known case and from several items of patient equipment. This led to a review of prevailing nursing practices that revealed that some items of respiratory equipment were being inappropriately decontaminated on one ward. These clinical procedures were subsequently modified.
Over the 6 months following detection of the index case, 256 staff screening samples were processed. No staff member was found to be colonised or infected with MDR-Ab.
During the outbreak, a total of 14 outbreak meetings with relevant staff were held. These meetings were chaired by the IPC doctor/consultant microbiologist and attended by representatives from the IPC team, occupational health, doctors, general managers, the director of nursing, staff from the affected wards, domestic services and PHE. At the point when wider screening was implemented, a brief was prepared to enable staff on the affected wards to explain the situation to patients. The IPC team members were on the affected wards every day during the outbreak and patients were given a contact card with the team's details if they required further information. Patients who were discharged before screening results were available were contacted by post. The letter contained screening results and advised who to contact if further information was required. Regular communication with staff was maintained through ward meetings and the regular and frequent presence of the IPC team.
Discussion
The outbreak lasted 207 days from the identification of the index case to the declaration of closure. MDR-Ab is one of the six most important MDROs in hospitals worldwide (Antunes et al, 2014). Hospital patients generally acquire MDR-Ab from contaminated equipment or by direct contact with colonised healthcare personnel (Nowak and Paluchowska, 2016). This trust's experience confirmed that MDR-Ab readily contaminates the environment and equipment.
Some screening protocols potentially underestimate the prevalence of MDR-Ab. The sensitivity of detecting MDR-Ab carriage based on culturing a sample taken from a single body site is probably less than 30% and sampling multiple sites is probably associated with a sensitivity of detecting MDR-Ab carriers of about 55% (Marchaim et al, 2007). The resurgence of the infection in the index case following a negative test after decolonisation may have reflected self-colonisation from a persistently positive body site or device. However, based on multiple testing at several body sites, it is the authors' view that most prevalent cases of MDR-Ab in the two adjoining wards were detected.
The authors believe this experience illustrates how readily MDR-Ab can spread. The index patient's cough would have made the pathogen highly infectious to patients and staff, with inevitable contamination of the environment. MDR-Ab proved very difficult to clear in the index patient. Therefore, management of the index case and other patients was aimed towards reducing levels of the pathogen. The octenidine decolonisation regimen, with antibiotics for those patients with an overt clinical infection, had some success in eradicating MDR-Ab. Decolonisation of all confirmed cases started in May 2015 and continued for the duration of the outbreak.
Measures were implemented in line with published guidance available at the time of the outbreak for the management of MDR-Ab outbreaks (Cookson et al, 2006). Microbiological testing showed that octenidine, which the trust was already using to decolonise MRSA-positive patients, was effective against MDR-Ab. In laboratory conditions, octenidine is more potent than chlorhexidine against, for example, Staphylococcus aureus, Escherichia coli, Klebsiella pneumoniae, Proteus mirabilis and Pseudomonas aeruginosa (Koburger et al, 2010; Spencer et al, 2013). Also, octenidine may be less likely to cause adverse effects (such as skin reactions) than chlorhexidine (Harris et al, 2015), although direct comparative studies are needed. Octenidine does not seem to have a detrimental effect on cells, such as fibroblasts and keratinocytes (Müller and Kramer, 2008), which might be relevant to wound healing and the maintenance of PEG and invasive lines. In contrast to chlorhexidine and mupirocin, there are no reports of resistance to octenidine. In vitro (test tube) exposure failed to induce octenidine resistance in MRSA (Al-Doori et al, 2007).
Multidisciplinary teamwork coupled with access to high-end, on-site decontamination processes plus the implementation of established barrier and hand-hygiene precautions, together with novel interventions, including the use of octenidine-containing products, may have worked synergistically and were critical to the management process. There is, however, a pressing need for further studies and consensus guidelines for the management of outbreaks of this potentially serious pathogen. Based on this experience, future consideration could also be given to the revision of multidrug-resistant Gram-negative bacteria guidelines to include octenidine as well as chlorhexidine. Since the outbreak the World Health Organization (WHO) has published guidelines that provide more up-to-date information regarding the management of MDROs (WHO, 2017).
Following reflection on events, experiences and the findings of the outbreak investigation, there is now an ongoing drive through mandatory IPC training, to educate and emphasise to staff the risks posed by MDROs and the associated IPC measures required. Management of the outbreak, including daily visits to the affected clinical areas by an IPC nurse, offered valuable opportunities to observe and reflect on clinical practice. This facilitated mutual learning and timely implementation of changes in practice (where appropriate).
In 2016 learning was shared internally and, more formally, by a conference/study day held for staff which was devised and delivered by IPC nurses alongside colleagues from other disciplines. Entitled ‘Multidrug-resistant Acinetobacter baumannii—our story’, the day covered not only antimicrobial resistance and a description of what happened during the outbreak, but also gave a laboratory perspective, advice on how MDROs might be managed in the community and interactive sessions on correct decolonisation and patient equipment cleaning techniques. Before this event, staff working on the affected wards had been invited to contribute to an audio recording charting their experiences of the outbreak. Several were keen to do so and these audio accounts were played on the day, followed by enthusiastic discussion.
The authors also engaged in wider, external sharing which included a poster presentation at the Infection Prevention Society (IPS) annual conference in September 2016 and participation as speakers at the PHE North East ‘Learning from Outbreaks and Incidents’ event in November 2016 and the IPS North East Branch conference held in June 2017.
Such sharing of this experience and lessons learned is vital if outbreaks of MDROs are to be contained successfully.

