The aim of this article is to critically discuss the aetiology of psychosis with the emphasis on genome research findings and the implications for this illness. Causal factors of psychosis have been controversial for many decades and countless explanatory models have been advanced by authors as a rationale for the illness: spiritual emergency (Crowley, 2006), stress vulnerability model (Zubin and Spring, 1977; Ursini et al, 2011), trauma (van Winkel et al, 2013), infections (Wockner et al, 2014), in utero (Uher, 2014), ethnicity (Korver et al, 2012; Schwartz and Blankenship, 2014), migration (Pinto et al, 2008), dopamine hypothesis (Tost et al, 2010; Melka et al, 2013), cannabis (French et al, 2015), heritability-environmental-risk (Plomin, 1994; van Nierop et al, 2013), neurodevelopmental/degeneration, genetics (Wockner et al, 2014), to cite a few. Bearing this in mind, although the trigger for symptoms of psychosis to develop has been theoretically postulated, the mechanism behind the trigger for the biological reaction remained speculative.
This curiously barren area of knowledge has come under the spotlight with the advent of genome mapping and the discovery of candidate genes and gene–environment interactions (Kukshal et al, 2012). This has led to rapid advances in genomic enlightenment regarding psychosis (Sagoo et al, 2009; McCarthy et al, 2014; Rees et al, 2015). These revelations have been supported and facilitated by an abundance of genome-wide association studies (GWAS) in search of the neuro-biological basis of psychosis (Corvin and Craddock, 2010; Valiente et al, 2011; Morgan et al, 2016). Much like the search for the ‘philosophers’ stone', this is not a new quest and much has been reported previously (Bilder et al, 2004; Sullivan et al, 2012; Harrison, 2015); what is new is the availability of new biotechnologies, which provide an interesting dilemma for ethical considerations, as the study participants, in the main, comprise genetic samples (Gejman et al, 2010; Clarke, 2014; Daca-Roszac et al, 2015; Morgan et al, 2016).
Historic to modern-day aetiology
The advent of Kraeplinian philosophy in 1911 divided mental illness according to two disparate diagnostic criteria; that of dementia praecox—typified by a youthful onset of florid symptoms: hallucination delusions, paranoia, marked by profound cognitive and functional impairment—and manic-depressive insanity: a cyclical pattern of intense mood swings. Kraeplin believed the origins of these illnesses were based in pathological diseases such as syphilis and tuberculosis. In 1919 Bleuler, a contemporary of Kraeplin, replaced the label dementia praecox with the familiar term of schizophrenia, forming the view that the florid symptoms were due to psychological deficits rather than Kraeplin's neuro-pathological model (Craddock and Owen, 2010). However, neither the pathological nor psychological models were able to explain the variation in degrees of presentation within schizophrenic (psychosis) indicators, for instance, the interactions and coexistence of positive (hallucinations, delusions, paranoia) and negative symptoms (cognitive impairments, lack of volition, poverty of thought) (Morrison et al, 2004; Reed, 2008), and a drop in functioning (work, relationships, education) (Owashi et al, 2009; Lyne et al, 2012). This lack of clarity lead to diagnostic and treatment uncertainty, alleviated by the development of the International Classification of Diseases (ICD) (World Health Organization (WHO), 2000) and later the Diagnostic and Statistical Manual (DSM) (American Psychiatric Association, 2013) (Tyrer, 2014).
Nevertheless, research continued to focus on a symptomology-driven diagnosis and the effects of psychosis on the person and society (Owen et al, 2007; Stafford et al, 2013; Thornton, 2016). Research of this type can be simplistically grouped under the broad heading of the nature–nurture debate or modernistically gene–environment interactions (Plomin, 1994; Boyd et al, 2013; Haworth and Davis, 2014). This century-old argument about that aetiology of psychosis has produced a rich classification of symptom-based publications on psychological and biological theories, which have impacted on clinical practice and public perception of this devastating mental illness (van Os et al, 2009; Boyd et al, 2013; Ronald, 2015; de Wit et al, 2016). Although psychosis is reported to occur in over 1% of the world's population (Saha et al, 2005; Dhindsa and Goldstein, 2016) there is little accord between these different factions, even to the point that the most research papers use the terms ‘psychosis’ and ‘schizophrenia’ interchangeably without acknowledging the advent of the psychosis continuum (van Os et al, 2009). For consistency, this article will use the term psychosis.
Psychosis and magnetic resonance imaging
Psychosis is considered a heritable disease of the brain with an inferred risk of vulnerability (Schlosser et al 2012; Light et al, 2014; Rees et al, 2015). Over time, familial vulnerability to psychosis has been established by twin studies revealing that relatives of those with psychosis are at 5-10 times higher risk of also developing psychosis than the general population (Korver et al, 2012). Indeed, research has shown similar anatomical brain incongruities within first-degree relatives sharing a 50% genetic link to the ill relative ie smaller hippocampal volume when compared with the study controls, and similar cognitive issues (MacDonald et al, 2009; Iveleva et al 2013). It should be noted that magnetic resonance imaging (MRI) scans from as early as 1999 have recorded structural brain alterations in psychosis, yet the rationale for causality and treatment has yet to be fully fleshed out (Harrison, 1999; Olabi et al, 2011; Vijayakumar et al, 2016). A comprehensive meta-analysis by Haijma et al (2013) found that loss of grey matter (frontal and temporal lobes, caudate nucleus and thalamus) increased with duration of illness. It is of interest that brain volume reaches 90% at age 5 years and completion at 14 years, according to Haijma et al, who hypothesised that brain volume reduction must be present when the cranium reaches maturation in early adolescence. This potentially links brain volume and psychosis to a combination of early neurodevelopmental issues and disease progression. Many researchers have pondered this conundrum for decades, giving rise to a plethora of in-depth research articles that cannot agree or state the origins for these brain differences and psychosis, apart from affirming that psychosis is a devastating brain disease and alterations to brain structure the visible result (MacDonald et al, 2009; Olabi et al, 2011).
Heritability and endophenotype
Contemporary genetic research has led to the understanding that the genome is the total collection of genes that defines an organism's genotype and phenotype (dog, cat, human etc) with epigenetic variations (colour, size, traits) due to environmental influences on heritable gene methylations (Lui et al, 2011; Modinos et al, 2013; Zhi et al, 2013; Light et al, 2014; Orgogozo et al, 2015). At this point it needs to be clarified that heritability of family characteristics is not a fact, it is a potential, and a descriptive risk statistic (Plomin et al, 1997; Haworth and Davis, 2014). The endophenotype is dependent on Mendelian recessive/dominant and gene penetrance for phenotypic expression linked to normal methylation and encountered environmental interactions (Lim and Maher, 2010; Jin et al, 2011; Heyn et al, 2013). Moreover, any two people can differ ad infinitum for expressed phenotype as research has confirmed that even monozygotic twins (commonly called ‘identical’ twins) are not totally identical (Castellani et al, 2015). Along with the awareness that interactions between the environment and genome impact on visible behaviours, a more complex picture is emerging with the classification of candidate genes ie COMT (Ira et al, 2013), DRD2 (Harrison, 2015), DSCI1(Gejman et al, 2010), which now includes a minimum of 108 associated interacting gene loci for psychosis (Schizophrenia Working Group of the Psychiatric Genomics Consortium, 2014). Therefore, psychosis cannot remain Mendelian in construct, but must be considered as a polygenetic heritability risk that falls within the domain of molecular genetics research (McCarthy et al, 2014; Rees et al, 2015). Even so, some fields of research find it difficult to acknowledge that psychosis could originate within hereditary risk factors, becoming verbose in their criticisms, citing poor study replication outcomes, specifically related to heritability estimates and gene risk (Light et al, 2014; Hall et al, 2015; Fosse et al, 2016). As a consequence of these concerns, which question the validity of classical twin studies and how genetics and environmental factors impact on study results, a literature search to develop a more in-depth awareness of polygenetic heritability and the link to the aetiology of psychosis, was implemented.
Systematic literature search
The aim of this systematic literature search was to identify genome studies and other articles pertinent to the purpose of this discussion (Boudha, 2010). The electronic databases PsychINFO, Medline and CINAHL were accessed (November 2016) and the following search terms used: Psychosis OR Schizophrenia AND Genetics OR Behavioural-Genetics OR Molecular Genetics, Heritability OR Inheritance, Aetiology OR Etiology. This returned a high number of hits, so a restriction to the search dates was applied (2013-2016) (Figure 1). Based on the hierarchy of evidence a title and abstract sift for exclusions was completed (Petticrew and Roberts, 2006). Twenty-nine full text articles were ordered. Those articles meeting the inclusion criteria defined by search terms were evaluated for quality of information. The database searches were extended by exploration of reference lists from full text articles, including relevant books and chapters (Horsley et al, 2011). A data extraction template was developed and key information from identified articles was tabulated. Quality was assessed by considerations of reliability, validity and bias of the study plus subject relevance.
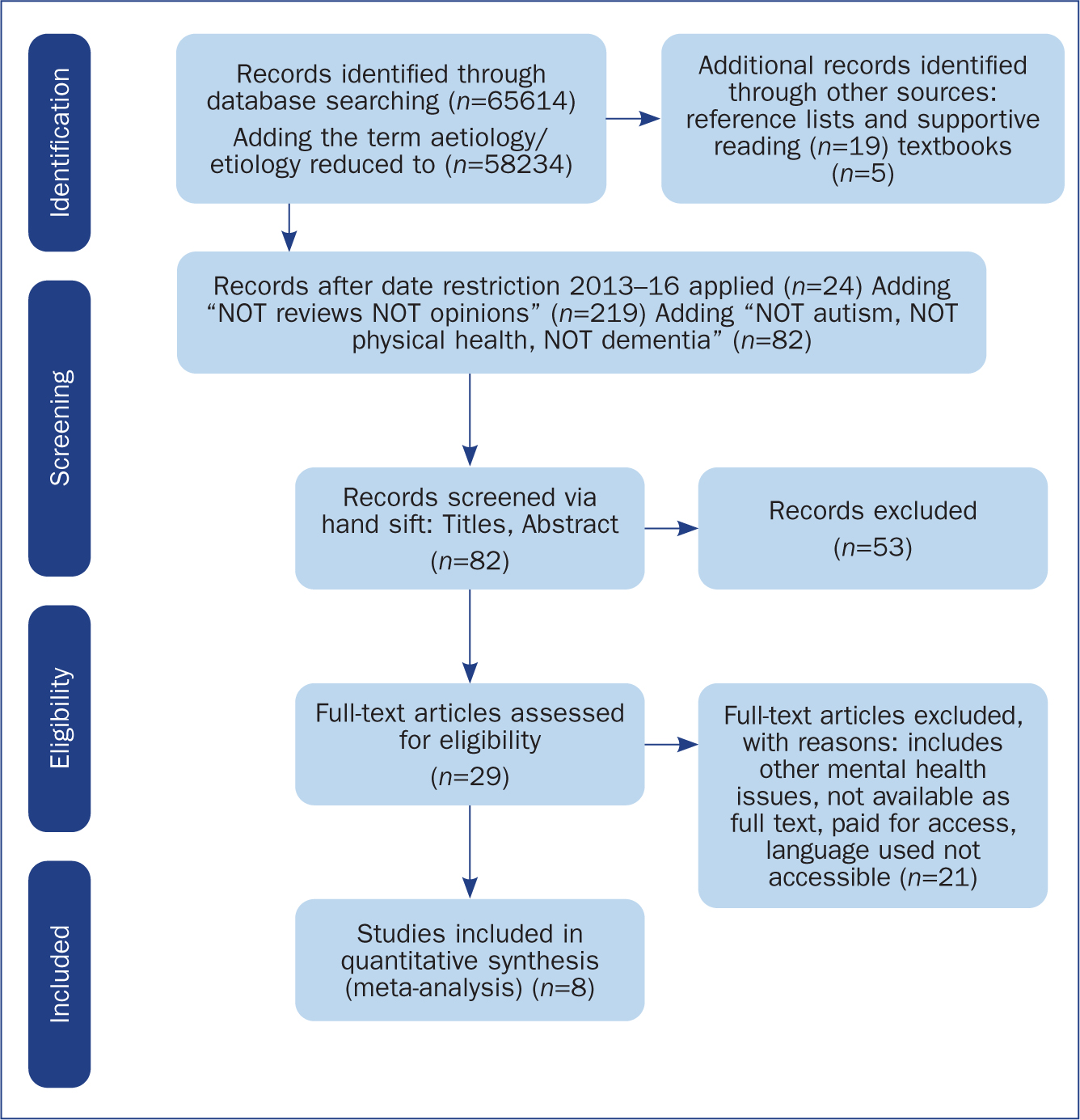
Methodology analysis
PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analysis) methodology (Shamseer et al, 2015) provides axiomatic appeal and is not alone in highlighting the need for consistency in reporting the method and the results sections, (indeed all sections) of the study published as a research article; specifically when the overall aim is to provide new knowledge based on reliable, valid and generalisable findings. It is with this intent that the eight identified articles were subjected to critical analysis to ascertain the quality of the studies. The eight articles were: Ira et al, 2013; Schizophrenia Working Group of the Psychiatric Genomics Consortium, 2014; Clarke 2014; Wockner et al 2014; Zammit et al, 2014; Zavos et al, 2014; Castellani et al 2015; Sekar et al 2016.
Quality assessment of genome articles
Initial quality assessment of the articles deemed suitable for this study using the PRISMA statement and checklist (Moher et al, 2009) identified only three studies of quality. Many of the articles appeared to be narrative in nature and difficult to identify as a case study, cohort, case-controlled, qualitative, quantitative etc. There appeared to be some form of dichotomy occurring, as the selected articles were published by reputable journals. Nevertheless those articles assessed against PRISMA guidelines were deemed of poor quality in all areas—absence of information, the methods section in the appendix, no method section (Table 1).
| Article type | Authors | Journal | Article layout |
|---|---|---|---|
| Case control | Castellani et al (2015) | BMC Medical Genetics | Abstract, background, method, results, discussion |
| Case control | Sekar et al (2016) | Nature | Abstract, introduction, results, discussion, extended data |
| Narrative, potentially a review | Schizophrenia Working Group of the Psychiatric Genomics Consortium (2014) | Nature | Abstract, introduction, discussion |
| Case controlled | Heyn at al (2013) | Genome Research | Abstract, introduction, results, discussion methods |
| Research investigation | Daca-Roszac et al (2015) | BMC Genomics | Abstract, introduction, results, discussion, methods |
| Unknown, potentially a review | Zhi et al (2013) | Epigenetics | Abstract, integrated introduction methods, results and discussion, no separation or headings |
However, this dichotomy was reconsidered and a potential mismatch between the quality assessment tools and the unique nature of the studies was considered a probable cause for the poor outcomes. Re-examining the reference section of the chosen articles (Horsley et al, 2011) three methodology articles were discovered, which provided information about a genetic quality assessment tool, STREGA2 (Strengthening the Reporting of Genetic Association Studies 2) (Hudson and Cooper, 2009; Little et al, 2009; Vandenbroucke, 2009). Furthermore, according to Sullivan et al (2012) genetic articles fell into three categories: narrative, case controlled or pedigree studies. Notably, Corvin et al (2010) emphasised that GWAS was an agreed, robust, valid and reliable methodological process, used in conjunction with statistical analysis, particularly logistic regression. All single nucleotide polymorphisms (SNPs) used in GWAS studies were assessed by experts in the field for quality and furthermore, statistical rigour was unyielding. GWAS ‘meets the community standards in human genetics for significance and replication’ (Sullivan et al, 2012). This information aided in the belated recognition that articles categorised as poor quality according to PRISMA tools were most probably within this specialist domain, a form of systematic review or meta-analysis. To ensure impartially, the critical analysis took two forms. First, tabularisation of the eight remaining articles. The detailed analysis of these eight articles identified issues with several articles such as missing methods section, biases, statistical analyse inconsistencies, validity and reliability considerations (the full analysis is available on request). Four articles were then randomly selected from the eight papers (using a coin toss for yes/no), for a narrative report.
Bearing this in mind and being unable to access the STREGA checklist, this author revisited the quality assessment tools and based on simplicity and clear explanation of use versus the complexity of genetic articles, the Joanna Briggs Institute quality assessment tools were deemed appropriate, as they included an opinion assessment to allow for narrative reports (Joanna Briggs Institute, 2020). This consideration is important as van Os et al (2009) highlighted the issues related to methodological uncertainties within genome/environment research, while urging the uptake of the Cochrane systematic review process by the genome community. This argument was also supported by Sagoo et al (2009). Simplistically and alternatively, prospective articles could be peer reviewed by non-genomic specialists.
Narrative analysis of four articles
Twin studies
Twin studies have been cited as the gold standard for research into environmental, genetic interactions and complex mental health presentations such as psychosis. Indeed it was twin studies that promoted the molecular genetic search for the cause of psychosis (Gejman et al, 2010). Equally, identical twins experience social interactions and environmental factors at the same level and time, from zygote division, birth and developmental stages, including familial and social environmental factors and remain the greatest for similarities (Geoffroy et al, 2013). However, this does not mean that shared life events are perceived in an identical way (Scarr, 1992).
No.1—Zavos et al (2014)
Zavos et al (2014) published an original twin study on the aetiology of frequent psychotic experiences (PE) in adolescence. Although the abstract was very clear it did not fully represent the content of the published article. For instance, the authors did not state that The Early Development Study (TEDS) was the basis for choosing subject participation. Furthermore, the non-participating families were reported to have higher scores on childhood behaviour problems than the participating families. This potentially introduced bias, as behavioural issues in childhood can be an indicator for psychosis risk. It was not reported in this article whether the parents of the twins had mental health issues, which would have impacted on the heritability estimates of psychotic symptoms/experiences leading to skewed results. This oversight brings into question study validity as the authors discussed genetic correlations and additive genetic influences by statistical interpretations only. Ultimately the study context was poorly reported and the association with the gene-life-laboratory was omitted. However, they concluded the study provided support for a psychosis continuum and that heritability depends on the type of PE experienced, with higher risk for paranoia and lower risk for hallucinations.
No.2—Castellani et al (2015)
In contrast, Castellani et al's (2015) article on methylation differences in monozygotic (MZ) twins discordant for psychosis was constant for title, abstract and article content. The aim of this study was to demonstrate that DNA gene methylation increased with age, leading to a rationale for psychosis discordance in twins. Surprisingly, discordant MZ twins were found to be more similar at the DRD2 gene to each other than to their own twin. In this study one twinset were Caucasian females age 43 (discordant for 16 years) and Afro-American females age 53 (discordant for 31 years). Clear explanation of DNA collection and SNP analysis by the ARRAY Star laboratories was reported. Of interest to this article is that the de-novo risk findings were linked to environmental exposures, ie development, and age but not ethnicity. The Caucasian twins genome-wide results indicated 111 genetic methylation episodes (GME) the Afro-Caribbean twins, 303 GME, with a crossover of 27 GME. Castellani et al suggested that it may be networks (linkage), rather than specific genes, as two networks were identified as important, cell death and survival, and cellular movement and immune trafficking; both networks previously identified in the aetiology of psychosis.(Castellani et al, 2015). For more information on gene methylation, please see Figure 2.

Genetics
In contrast to twin studies, the advancement in technical arrays, analysis and collaborative collections of genetic samples has enabled co-operation in laboratory-based research, initially lead by GWAS, specifically for neuro-psychiatric illnesses such as psychosis. Daca-Roszac et al (2015), emphasised the need for meticulous analysis of array printouts, as it is reported that methylation test results could be impaired if the researchers have not pre-identified a SNP variation, as some of the arrays have technological differences.
No.3—Schizophrenia Working Group of the Psychiatric Genomics Consortium (2014)
According to this article, genotype single nucleotide polymorphisms (SNP) and copy number variants (CNV) are common areas of investigations and their effect on gene expression and allele deletion in relation to disease phenotype. These investigations expedited the discovery of complex polygenetic heritable variance in the aetiology of psychosis. To date 108 independent gene loci have been discovered on various chromosomes, specifically chromosome 6, and include DRD2 (dopamine) and CD20 (immune) genes. The SWGPGC (2014) narrative report pointed out that additional associations for psychosis are to be found within the important immune function associated with the major histocompatibility complex (MHC). It highlighted an interest in rare genetic variations (CNV) and considered these studies linked with biotechnology will provide more information about psychosis. This report was based on the work of potentially more than 100 researchers from different sites around the world.
No.4—Sekar et al (2016)
An article by Sekar et al (2016) reported on the MHC interaction with the complement component 4 (CC4) variant (found throughout neurons: axons synapse) that implies a high-risk indicator for psychosis. CC4 is considered an important component of in-place immune system pathways that eliminates cell debris and has been implicated in the facilitation of neuronal pruning of synapses in the brains of teenagers. This synaptic pruning, occurring until the age of 30, is considered the final period of cortical development. The authors' deliberation was that psychosis is associated with the MHC and allelic differences with the CC4 gene. Arrays were used to analyse genetic samples from this large study of 28 799 (psychosis) and 35 986 (control) SNPs and results were available for scrutiny.
Discussion
A constellation of research from the past decade into the origins of psychosis, specifically the genetic origins, has not as yet been perfected, mainly due to the arguments about effective methodology and interpretation of results. Sciences such as molecular genetics explore the world of minutiae within the biological structure of a cell, with tangible substances in the form of genetic samples, blood, saliva, tissue as their participant groups. Whereas the majority of research published about psychosis deals with human frailty, the inconsistencies, and nebulous structures that represent the symptoms of psychosis, and not the biological basis and the effect on this brain disease. Within the procedure-orientated world of molecular genetics, it is understandable that results and not methods have primacy. However, for generalisation and to strengthen reliability and lessen criticism of geneticists' important findings, research reporting will need to be standardised, along with genome sample demographic variables, which was highlighted as an issue in the article by Castellani et al.
One aspect of this article's question asked if modern research provided enlightenment into the understanding of the origins of psychosis. The simplest answer is yes. Despite the criticisms, the study of the human genome has brought new knowledge into the discernment of the aetiology of psychosis. It is now known that there are candidate genes for susceptibility to psychosis, which are located in microscopic regions of the brain—for instance, the major histocompatibility complex. Considering the polygenetic effects, genes and loci interact with each other to provide heritable vulnerability patterns within families. It was known before the GWAS studies that there was a risk bias within a family pedigree but not the neurobiological rationale for the risk. Also highlighted in the research cited in this article is the impact of the environment, which takes many diverse forms: in-utero, infection, cranium maturation; on genetic activity in the form of methylation processes, and the activation of de novo high-risk alleles. This reduces the argument of the nature or nurture debate, as the findings indicate rather a nature and nurture basis for de-novo psychosis (no family history). Specifically, when linked to symptoms associated with psychosis.
Subjectively it appears there may be two neuro-pathological pathways to the development of psychosis: familial risk heritability, and the stimulation of non-inherited de-novo risk alleles via environmental impact under the banner of epigenetics. Additionally, and of interest to the origins of psychosis is that of the MHC and neuronal differentiation linked to CC4 pruning in adolescence, which may be central to gene–environment interaction, while also hinting at a rationale for the reduced brain volume of people experiencing psychosis, noted in MRI scans. To discover the commonalities of environmental exposure could potentially be a new area of early intervention research. This new way of thinking provides a solid foundation for many of the evidence-based psychological theories such as the stress vulnerability model or trauma, as the de novo methylation event does not change the gene and is therefore potentially reversible. Ursini et al (2011) posited that environmental stress and COMT interaction (by SNP-methylation) can impact on frontal lobe function; again providing support for epigenetics and gene environment interactions. Not to be forgotten is the dopamine hypothesis, which can now be linked to the activity DRD2 gene and associated receptors, when interacting with COMT allele, which is responsible for dopamine degradation and flux (Tost et al 2010). This flux can have a negative impact on cognitive functions i.e. executive activity and memory. The dopamine hypothesis still remains, as do the psychological theories; there is now a strong neuro-biological basis to its premise. However, Wockner et al (2014) cautioned that some DNA methylation events in brain tissue are different from that in blood or saliva samples, which may account for differences in reported research findings.
Conclusion
In the introduction this author referred to the philosophers' stone; alchemists of bygone eras would probably consider many of the scientific developments of today the stone's actualisation. Comparably, without the advent of array bio-technologies for analysing and collecting genetic samples, the minute differences in DNA caused by SNP-associated methylations and high-risk alleles would not have been visible to earlier forms of laboratory equipment. Therefore, the new developments in understanding the cause of serious mental health illnesses, such as psychosis, would not be written, nor would the hope of finding a way to lessen the devastating effects of this illness be so promising. This may mean designer medications to target specific genes, loci or even altering the operational diagnosis of psychosis by subjectively reported symptoms, replacing them with on-the-spot genetic sampling and analysis. This does not preclude advances in psychological theories related to epigenetic effects on behaviours; rather it opens new vistas of exploration potential in the domain of negative psychosis symptoms. However, to continue to move forward requires ethical co-operation and exchange of evidence-based theory, not just within the field of genetics but also with the softer sciences, for the benefit of individuals and families experiencing psychosis.

